COVID Jabs Have Been in Research Since 1960 - Under the Guise of Rare Diseases Targeted with CAR T Autologous Therapies
Tune in and hear the truth...
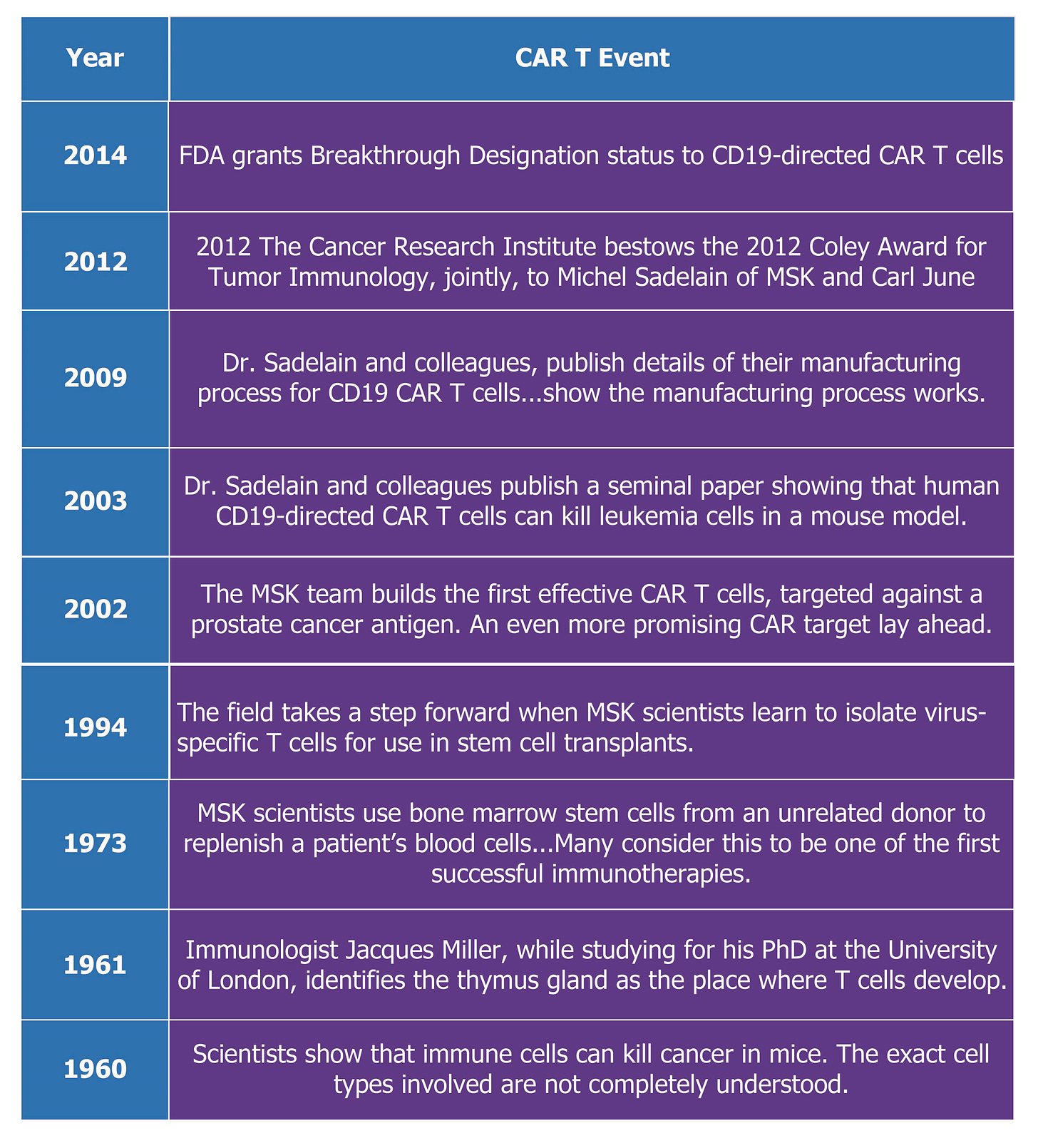
CAR T Therapies for Rare Diseases Have Been in Development Since 1960
Since 1960, research has been underway in a category of biologic products known as chimeric antigen receptor T (CAR T) therapy.
The table below shows the key milestones selected from the timeline in discovering and developing a CAR T therapy:
The history is summarized on the Memorial Sloan Kettering Cancer Center website titled CAR T Cells: Timeline of Progress.
Is this much needed pioneering research from Memorial Sloan Kettering Cancer Center Memorial Sloan Kettering Cancer Center?
Or is it fast-tracking a therapy based on scant evidence that a safe manufacturing process had been devised?
This is taken from the MSK full account of the timeline:
“2009
Dr. Sadelain and colleagues, including Isabelle Rivière, publish details of their manufacturing process for CD19 CAR T cells, to be used in patients with relapsed, chemorefractory leukemia. They show that the manufacturing process works and the cells are effective.”
Are you in a Deep State of Shock Now?
If you aren’t in shock, you should be.
A few scientists in a laboratory claimed they could ‘manufacture’ these therapies. If they were right, then my name is Donald Duck!
The first of these CAR T therapies was approved by FDA in August 2017, know as Kymriah. This is the package insert with an FDA Black Box warning on it:
HIGHLIGHTS OF PRESCRIBING INFORMATION (Click to see full details)
“WARNING: CYTOKINE RELEASE SYNDROME, NEUROLOGICAL TOXICITIES, and SECONDARY HEMATOLOGICAL MALIGNANCIES”
None of the CAR T therapies (around 9) were selling, so they rebranded them as vaccines!!!
I’ve Provided Evidence to a Number of Lawyers, including Aaron Siri’s team, to no Avail
Lawyers and attorneys have not picked up on the evidence I have, disappointingly.
Subscriber Phillip Bartleman asked me about it last week, suggesting I contact Aaron Siri, to which I responded that I had calls and email exchanges with his senior partner, Elizabeth Brehm, beginning August 2021. That was in response to my message to ICANs website. This is the text:
“Dear ICAN,
I am an expert in biopharmaceutical supply chains – please see my resume attached.
Also, I have a long history as a campaigner for wholesale reform of Big Pharma, and am well known as an industry insider who speaks truth to power.
There is so much people don’t know about the supply chains for these vaccines. Quoting from my latest book – “Pharmaceutical supply chains produce the drugs that enter a patient’s body. If anything goes wrong in production, even though the Regulatory Authority has approved a drug for sale, it can have a devastating impact.”
These are the things people don’t seem to know:
None of the 4 vaccine ‘manufacturers’ developed or manufactured the vaccines themselves. None of the manufacturers have ever had a vaccine as part of their product portfolio, so they could be deemed as lacking sufficient experience to sponsor vaccine development and manufacture.
For Pfizer/BioNTech and Moderna mRNA vaccines, Lonza manufacture the bulk drug substance and ship to Catalent Pharma Solutions for the fill/finish.
For AZ/University of Oxford, Oxford BioMedica developed and manufacture the adenovirus bulk drug substance, and Wockhardt carryout fill/finish.
For Janssen (J&J)’s adenovirus, it is the same contract manufacturers as Pfizer/BioNTech and Moderna.
The manufacturers are legally responsible for the work of the contract manufacturing companies, according to CGMP, which is included in the US Code of Federal Regulations (CFR).
FDA’s portion of the CFR is in Title 21, which interprets the Federal Food, Drug and Cosmetic Act and related statutes, including the Public Health Service Act.
The main elements of CGMP are: 21 CFR Part 210 Current Good Manufacturing Practice in Manufacturing Processing, packing, or Holding of Drugs. 21 CFR Part 211 Current Good Manufacturing Practice for Finished Pharmaceuticals. 21 CFR Part 600. Biological Products: General.
In my expert view, it is physically impossible to develop vaccines on the timescales they have been, without contravening major elements of CGMP, such as process validation, audit of suppliers, change control, etc, etc.
I personally spent 6 months consulting at Oxford BioMedica on the Lentivirus vector used in Novartis’ Kymriah CAR T gene therapy treatment for blood cancers and there is no doubt the vaccines are gene therapy.
Please let me know if I could help further in any way.
Kind regards,
Hedley
Hedley Rees
Managing Consultant and Author
T: +44 1656 655664
M: +44 7734 961726
Book: Supply Chain Management in the Drug Industry: Delivering Patient Value for Pharmaceuticals and Biologics - Read Me
Book: Taming the BIG PHARMA MONSTER by Speaking Truth to Power – Read me
Book: What Patients Need to Know About Pharmaceutical Supply Chains – Read”
I had a few calls with Ms Brehm at the time, and sent her a lot of evidence, but nothing further transpired.
I’m still open to provide the evidence if so required.
Hedley


65 years later they still have no clue what the heck they are doing. I wouldn't trust these bozos with putting a band aid on a cut.
Thank you Ms Rees! You are doing what very few others hold the qualifications for; holding expert, truthful, and unequivocal facts out in the Light as a source of truth for all the rest of us. Please, don’t ever give up! Thank you ! I’m so much better informed now than I was before reading your post. Keep it coming ! 🙂